Mechanical forces orchestrate the epigenetic landscape of oral mesenchymal stem/progenitor cell fate in dental and periodontal tissues
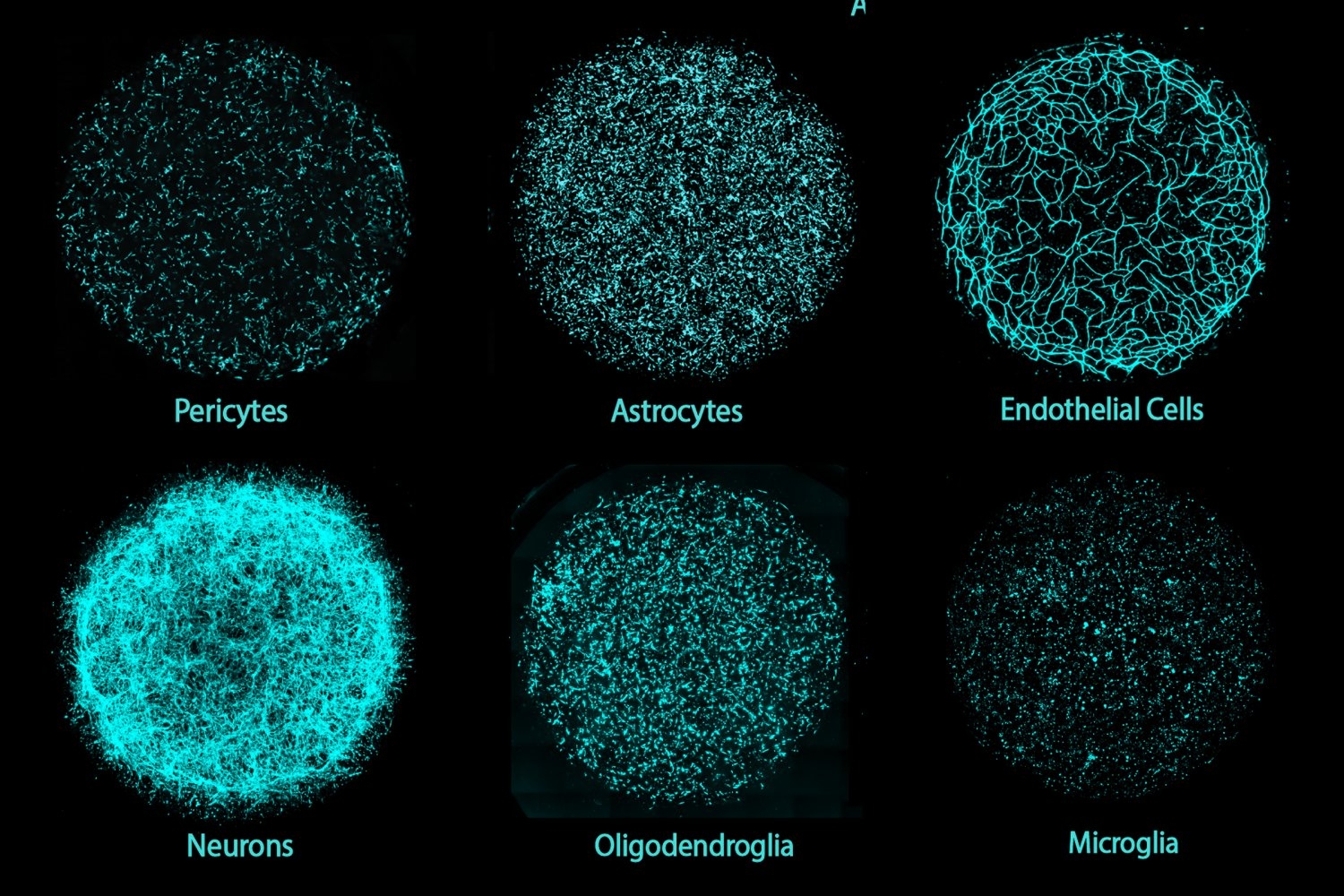
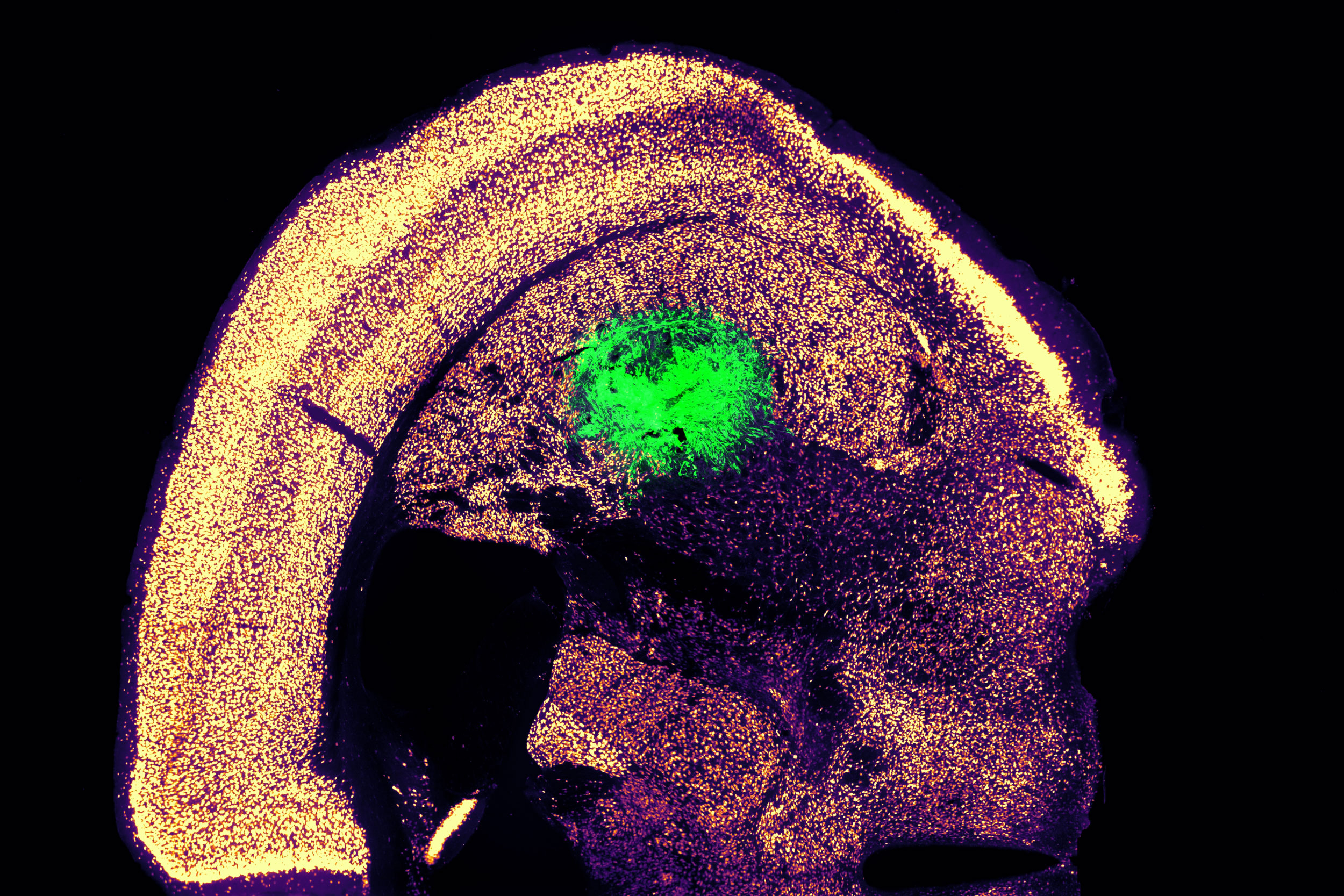
The oral cavity serves as the primary source of oral mesenchymal stem/progenitor cell populations residing in the dental pulp, periodontal ligament, deciduous tooth pulp, and gingival connective tissue. Oral and periodontal tissues exist in a constantly loaded biomechanical environment, where forces from mastication, vascular pulsation, and orthodontic manipulation continuously act on resident mesenchymal stem cells, including dental pulp stem cells (DPSCs), periodontal ligament stem cells (PDLSCs), stem cells from human exfoliated deciduous teeth (SHEDs), and gingival mesenchymal stem cells (GMSCs). In this review, we use the term “oral stem cells” to specifically denote oral mesenchymal stem/progenitor populations residing in dental pulp, periodontal ligament (PDL), deciduous tooth pulp, and gingival connective tissue (DPSCs, PDLSCs, SHEDs, and GMSCs), which are most relevant to orthodontic remodeling and dento-periodontal regeneration. For clarity, this review highlights the defining characteristics, representative markers, differentiation potential, and immunomodulatory properties of these oral stem cells within the manuscript, establishing a foundation for understanding how mechanical forces shape their fate. These forces are not merely physical stimuli; they actively reshape stem cell fate by engaging a multilayered mechano - epigenetic regulatory network that integrates cytoskeletal mechanotransduction, nuclear mechanics, and chromatin remodeling. Mechanical inputs such as compression, tension, shear stress, and extracellular matrix stiffness modulate DNA methylation, histone acetylation and methylation, 3D genome architecture, and non-coding RNA programs. These epigenetic and epitranscriptomic adaptations stabilize lineage commitment, influence inflammatory and regenerative outputs, and may establish “mechanical memory” that persists after load removal. Metabolic rewiring, including YAP/TAZ- and MAPK-driven control of mitochondrial activity and metabolite pools, provides an additional axis linking mechanics to chromatin state. Building on these mechanisms, emerging therapeutic strategies aim to couple defined mechanical cues with epigenetic modulators and mechano-tunable biomaterials to enhance pulp regeneration, periodontal repair, and orthodontic bone remodeling with higher precision. The review further highlights single-cell multi-omics and live-cell imaging approaches as essential tools to resolve force-dependent chromatin dynamics in vivo, and proposes that integrating biomechanics, epigenetics, and metabolic control will enable next-generation regenerative dentistry and personalized orthodontic intervention.